Introduction to Muscle Contraction, Part 1
Atomic Structures of Actin and Myosin
Actin (thin filament) fibres are helical polymers of g-actin (globular-actin) . The structure of monomeric actin which contains 365 residues and has a molecular weight of 42,000 was solved by protein crystallography as a 1:1 complex with the enzyme DNase I (Kabsch et al. 1990). Orientated gels of actin fibres (f-actin), a helical copolymer of actin which has 13 molecules in 6 turns repeating every 360Å, yield x-ray fibre diagrams to about 6Å resolution. It was possible to determine the orientation of the g-actin monomer which best accounted for the f-actin fibre diagram (Holmes et al. 1990) and thus arrive at an atomic model of the actin filament (Lorenz et al. 1993).
© Kenneth C. Holmes
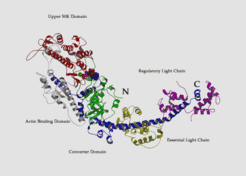
The cross-bridges comprise a part of the myosin molecule, namely subfragment-1 of heavy meromyosin (S1). The structure of chicken S1 has been solved by x-ray crystallography (Rayment et al. 1993) - in the following references to residues in the chicken structure will be prefaced with "gg". This study shows the S1 (which has 884 residues) to be tadpole-like in form, with an elongated head, containing a 7-stranded β sheet and numerous associated a-helices forming a deep cleft, with the actin binding sites and nucleotide binding sites on opposite sides of the sheet. The cleft separates two parts of the molecule which are referred to as the 50K upper and 50K lower domains or actin binding domain.
© Kenneth C. Holmes
The cross-bridges comprise a part of the myosin molecule, namely subfragment-1 of heavy meromyosin (S1). The structure of chicken S1 has been solved by x-ray crystallography (Rayment et al. 1993) - in the following references to residues in the chicken structure will be prefaced with "gg". This study shows the S1 (which has 884 residues) to be tadpole-like in form, with an elongated head, containing a 7-stranded β sheet and numerous associated α-helices forming a deep cleft, with the actin binding sites and nucleotide binding sites on opposite sides of the sheet. The cleft separates two parts of the molecule which are referred to as the 50K upper and 50K lower domains or actin binding domain.
The C-terminal tail, sometimes called the neck, which also provides the connection to the thick filament forms an extended α-helix which binds two light chains. The ATP binding site contained the typical P-loop motive which is also found in the G-proteins.
© Kenneth C. Holmes
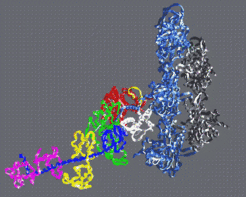
Fig 4: By fitting the atomic structures of f-actin and S1 into three dimensional cryo-electron microscope reconstructions one arrives at an atomic model of the actin myosin complex (Rayment et al. 1993).
© Kenneth C. Holmes
By fitting the atomic structures of f-actin and S1 into three dimensional cryo-electron microscope reconstructions one arrives at an atomic model of the actin myosin complex (Rayment et al. 1993, Fig 4). In particular, this model establishes the spatial orientation of the S1 myosin fragment in the active complex. For example one finds that the cleft in myosin extends from the ATP binding site to the actin binding site and that the opening and closing of this cleft is very likely to provide the communication between the ATP site and the actin binding site. The actin binding site spans the 50K upper and lower domains and the ATP binding site extends from the 50K upper domain into the 50K lower domain. Furthermore, the very extended C-terminal α-helical neck of S1 is ideally placed to be a lever arm. The lever arms joins onto the bulk of the molecule via a small compact converter domain (Houdusse and Cohen 1996) which lies just distal to a broken α-helix containing two reactive thiol groups known as SH1 and SH2. Numerous experiments point to the putative hinge for the lever arm being in the SH1-SH2 region of the molecule (see Holmes 1997 for review).

