Biomimetic Hydrogels

Influence of Charge on Extracellular Matrix Properties
Hyaluronic acid (HA) is a vital, functional component of the extracellular matrix (ECM) in vertebrate tissues. It represents a highly charged and hydrated network in which different cells are embedded. Following a bottom up synthetic biology approach a minimal ECM mimetic with a controlled chemistry and variable charge is designed as a polyelectrolyte hydrogel. It is built up of HA as highly negatively charged polymer backbone and two types of short covalent crosslinkers, giving the opportunity to vary material properties by adjustment of the negative network charge. In a thiol-Michael addition defined network structures are created, which can be tuned by the degree of functionalization of the HA chain as well as the aromatic core and charge of the crosslinkers. This system is furthermore characterized for its charge related physico-chemical properties and bioactivity.
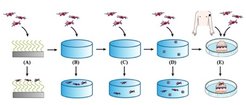
![Schematic representation of the architecture of HA hydrogels, with structural details for all important components and the influence of crosslinking on charge density[1,2].](/14217740/original-1537869438.jpg?t=eyJ3aWR0aCI6MjQ2LCJvYmpfaWQiOjE0MjE3NzQwfQ%3D%3D--4c1f7736c8995fea80acde204c1d87468f6449e8)
Schematic representation of the architecture of HA hydrogels, with structural details for all important components and the influence of crosslinking on charge density[1,2].
With increasing negative network charge the Young’s modulus of the hydrogels decreases linearly as swelling ratios and respective mesh sizes increase. These properties are based on the secondary interactions with the aromatic pyridinium-bisacrylamide-crosslinkers (1a and 1b). In respect to bioactivity the presented hydrogels show enzymatic degradability and cell attachment, dependent on both covalent and electrostatic interactions within the hydrogel network. Furthermore, no unspecific or specific attachment of proteins to these hydrogels is observed. In addition to fundamental insights in charge related ECM behavior, the influence of electrostatic properties could also lead to innovations in existing biomedical products.
[1] Hegger et al. ChemistrySelect, 2017, 25:7701.
[2] Hegger et al. Molecules, 2018, 23, 769.
All Hyaluronan Hydrogels
Hydrogels have proven useful in a range of cell culture applications, revealing fundamental phenomena regulating cell behavior. They provide new tools for the expansion and directed differentiation of various cell types virtually impossible with conventional culture substrates. Their resemblance to living tissue opens up many opportunities for application in biomedical areas. Currently, hydrogels are used for manufacturing contact lenses, hygiene products, tissue engineering scaffolds, drug delivery systems and wound dressings. Tissue engineering itself, has changed the therapeutic approach to tissue regeneration and replacement. The recent developments in this field come from reparative medicine in addition to the need for tissues and organs required for transplants. A remaining problem after the transplant of organs or tissues still is rejection of the transplanted organ or tissue by the patient´s organism. The ideal way to circumvent these rejections is to use the patient's own cells. An alternative approach are biodegradable materials that can promote the ingrowth of neighboring tissues and cells or serve as a provisional scaffold for transplanted cells to adhere, proliferate, and differentiate within.
Hyaluronan, together with its receptors and binding partners, is a “keystone molecule” in the inflammatory milieu. Meaning that HA is at the centre of a complex network of ECM molecules that together exert decisive effects on the nature of inflammation.
Following the bottom up approach of synthetic biology a hydrogel should be created using one singular component of the ECM in order to reduce complexity as well as adverse reactions by cells and organisms.

![Schematic representation of the architecture of HA hydrogels, with structural details for all important components and the influence of crosslinking on charge density[1,2]. Schematic representation of the architecture of HA hydrogels, with structural details for all important components and the influence of crosslinking on charge density[1,2].](/14217740/original-1537869438.jpg?t=eyJ3aWR0aCI6MzQxLCJmaWxlX2V4dGVuc2lvbiI6ImpwZyIsIm9ial9pZCI6MTQyMTc3NDB9--5897619e6409643675c595858ad78b939fb1cc8f)
