Enzyme skyscrapers help microbes grow
Scientists determine structure of an important nitrite-oxidizing enzyme in the global biological nitrogen cycle
Scientists from three Max Planck Institutes (MPI), the MPI for Medical Research in Heidelberg, the MPI for Marine Microbiology in Bremen and the MPI for Biophysics in Frankfurt and the Radboud University in Nijmegen have successfully determined the structure of the enzyme which produces a large part of the nitrate on earth. Microorganisms use this enzyme, called NXR or nitrite oxidoreductase, to convert toxic nitrite into nitrate. Both nitrite and nitrate are forms of nitrogen that have a major impact on our environment. The results were recently published in the journal Nature Microbiology.
Nitrate is a nutrient and an important molecule for many biological processes, for example for the growth of plants. It occurs in large quantities (approx. 600 billion tons) on earth. Almost all of the naturally occurring nitrate is produced by the oxidation of nitrite by the enzyme nitrite oxidoreductase (NXR). NXR is thus a crucial enzyme in the global biological nitrogen cycle. It occurs in nitrite-oxidizing bacteria (NOB, including comammox organisms), which produce most of the nitrate in the environment, as well as in anaerobic ammonium-oxidizing (anammox) bacteria. Anammox bacteria produce around half of the nitrous oxide (N2) in our atmosphere. The scientists involved have now been able to elucidate the structure of the enzyme NXR from anammox bacteria.
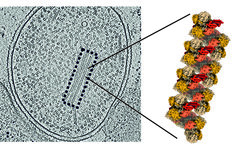
The results show in detail how NXR is structured: The individual NXR units are stacked like bones of the spinal column on top of each other, a structure that resembles a skyscraper has not been observed before. "Cryo-electron tomography and helical reconstruction have shown that NXR forms these unusual tubular structures, just like long fibers, in the anammox cells," says Kristian Parey from the Max Planck Institute for Biophysics. Their function is currently still unclear. The structure of the single NXR molecule is clearer: on one side there is a so-called “active pocket” in which nitrite is bound and converted into nitrate. Electrons are released and transported to the other side of the molecule via a kind of electrical line. These electrons can then be picked up by other molecules and used for cell metabolism.
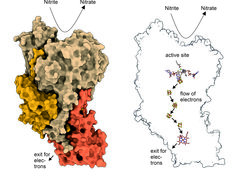
"Because of its importance for the global nitrogen cycle, NXR has been studied for decades, but we didn't know exactly what NXR molecules look like," says Thomas Barends, group leader at the MPI for Medical Research in Heidelberg. "Now we know the detailed structure of these molecules, which helps to understand how NXR works."
"In the case of anammox bacteria, the connection between nitrite oxidation and nitrite reduction was previously a mystery," adds Boran Kartal, group leader at the MPI for Marine Microbiology in Bremen. "This study takes us a big step further in understanding how these microorganisms combine energy production and growth." The anammox process is a particularly exciting part of the nitrogen cycle: this process converts nitrite or nitrogen oxide and ammonium directly into molecular nitrogen (N2), which accounts for 78% of our atmosphere. "Our detailed investigation of the molecular mechanism of nitrite oxidation will help us to uncover the factors that control the nitrate production in nature, which is an important step in the ecological nutrient cycle," continues Kartal.

“Due to the great environmental impact of nitrite and nitrate, it is important to know how bacteria handle these substances in order to learn from their processes,” Barends emphasizes. Because of their properties, anammox bacteria are regularly used to purify wastewater in sewage treatment plants. Increasing our understanding of how they function could lead to more efficient use in the future.


